 Poster presented at: the 11 th International Conference on Advanced Technologies & Treatments for Diabetes: February 16, 2018: Vienna, Austria. Comparison of flash glucose monitoring usage patterns and glycaemic outcomes in the real-world with those observed in a randomized controlled trial. Poster presented at: the 11 th International Conference on Advanced Technologies & Treatments for Diabetes: February 17, 2018: Vienna, Austria. Sustained improvement in glycaemic control following flash glucose monitoring: a worldwide observational analysis. Real-world flash glucose monitoring patterns and associations between self-monitoring frequency and glycaemic measures: a European analysis of over 60 million glucose tests. 
Flash glucose-sensing technology as a replacement for blood glucose monitoring for the management of insulin-treated type 2 diabetes: a multicenter, open-label randomized controlled trial. Dexcom and Medtronic are among the other big names in the industry in the U.S. Freestyle Libre Flash Glucose Monitoring System ( Reader & Sensor. 
In June, the med tech company got the FDA greenlight for its FreeStyle Libre 2 to be used by adults and kids over the age of 4 with diabetes.Ībbott has competition in the CGM market. Benefits for FreeStyle Libre 2 sensors apply to two FreeStyle Libre 2 sensors per. At the time, the system was cleared for 10-day use, but in 2018 Abbott got another clearance for its FreestyleLibre 14 Day Flash Glucose Monitoring System. In 2017 Abbott’s Freestyle Libre system became the first CGM to be FDA cleared for use without fingerstick. The Libre Sense was designed to help athletes understand the efficacy of their nutrition choices on training and competition. Earlier this month it announced that its Libre Sense Glucose Sport Biosensor earned a CE Mark. Within the last month Abbott has had its sights set on the European market. (106) Class I devices that are low-risk are subject only to general controls. "People living with diabetes are at the center of our design process, and we made our next-generation technology even more discreet for a better user experience to make managing diabetes as easy and seamless as possible." Keywords: CGM Continuous Glucose Monitoring FSL FreeStyle Libre. We've done that again with FreeStyle Libre 3, the smallest sensor that delivers life-changing benefits and best-in-class accuracy," said Jared Watkin, senior vice president for diabetes care at Abbott. Scottish Diabetes Group, the Association of British Clinical Diabetologists. "Abbott won't stop innovating when there's room to raise the bar. This guidance has considered the advice provided by the. The company is pitching this tool as a convenient way for those living with the disease to monitor it. 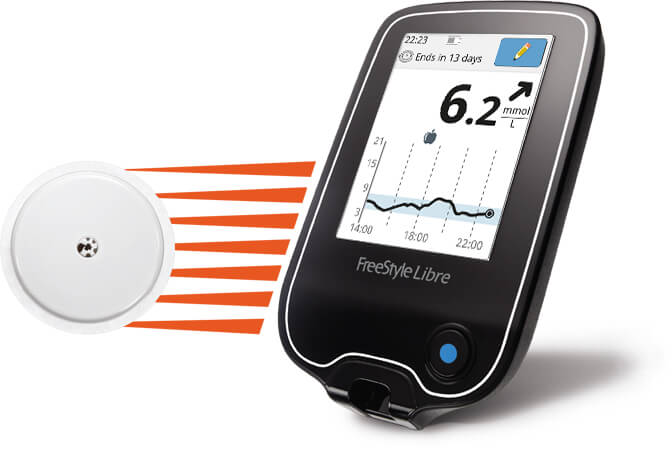
That number is growing, according to the agency, who attributes the uptick to obesity, unhealthy diet and physical inactivity. The company said that it will price the new product at the same price point as the last version of the device.Īccording to the World Health Organization, there are 60 million people in Europe with diabetes. While the technology is not yet available in the U.S., according to an Abbott spokesperson the company is planning to roll out the technology in other markets pending regulatory approval. Because the technology is smaller than previous versions, the company said that it is better for the environment. This new design marks the first time that the company has changed the design since launching the original FreeStyle Libre. The technology is also able to give users alerts and trends data via their smartphone app. Users apply the one-piece applicator to the back of the upper arm. This latest version is smaller than previous versions –about the size of two stacked U.S. from European countries with type 1 and 2 DM at 6 months post intervention. 
Like its predecessor this monitor can also last for 14 days and transmit glucose data to a users’ smartphone every minute. FreeStyle Libre Flash Glucose Self-Monitoring System: A Single-Technology. This news comes just a few months after the company landed the much-awaited U.S. This morning Abbott announced that its continuous glucose monitor the FreeStyle Libre 3 earned a CE mark, making it cleared for use for individuals with diabetes living in the European Union. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
